Ch. 12
i)

ii)

iii, iv)

acid catalyzed addition of H-OH (hydration) to alkene;
{hydration mechanism}
oxymercuration-demercuration
{hydration mechanism}
oxymercuration-demercuration
v)

vi)
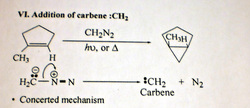
vii)

addition of Br2 and Cl2 (halogenation) to alkene
should have solvent CCl4
Wikipedia
Chemwiki
HALOHYDRIN (aka haloalcohol) formation 1 halogen, 1 OH
should have solvent CCl4
Wikipedia
Chemwiki
HALOHYDRIN (aka haloalcohol) formation 1 halogen, 1 OH
viii)
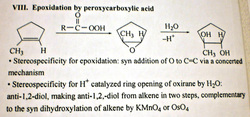
epoxide (aka oxacyclopropane) synthesis by peroxycarboxylic acid (peracid)
scroll down lower to see ANTI di-hydroxylation
(add 2 OH) --> anti di-ol
scroll down lower to see ANTI di-hydroxylation
(add 2 OH) --> anti di-ol
ix)

dihydroxylation by KMnO4 or OsO4
SYN di-hydroxylation --> syn di-ol
SYN di-hydroxylation --> syn di-ol
x, xi)
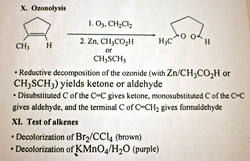
ozonolysis
test of alkenes
test of alkenes
