iii)

synthesis of alkynes by elimination reactions
good examples (see #4a!!)
<second example of geminal dihalide with t-Bu: should be dehydrohalogenated twice (2 NaNH2), not three times...?
good examples (see #4a!!)
<second example of geminal dihalide with t-Bu: should be dehydrohalogenated twice (2 NaNH2), not three times...?
iv)

synthesis of alkynes by alkylation of terminal alkynes
<alkynol = alkyne with OH group
note: terminal alkynes are more acidic
<alkynol = alkyne with OH group
note: terminal alkynes are more acidic
v)

reduction by catalytic hydrogenation
partial hydrogenation (alkyne --> alkene) CIS
partial hydrogenation (alkyne --> alkene) CIS
vi)

reduction by dissolving metals, aka partial hydrogenation
(alkyne --> alkene) TRANS
(alkyne --> alkene) TRANS
vii)
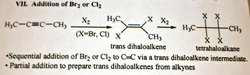
addition of Br2 or Cl2 (halogenation) to alkyne
(dihalogenation)
(dihalogenation)
viii)
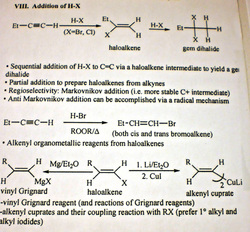
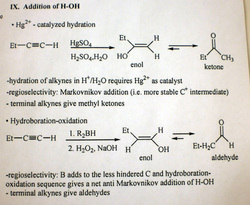
addition of H-X (hydrohalogenation) to alkyne
regular: Markovnikov
otherwise: anti-Markovnikov via Radical Addition mechanism (second half)
regular: Markovnikov
otherwise: anti-Markovnikov via Radical Addition mechanism (second half)
ix)